By Azer Parrocha
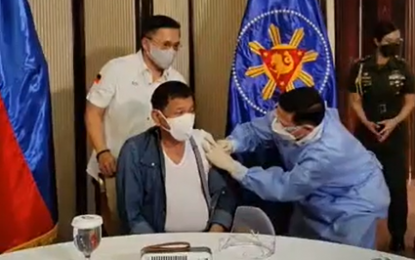
SINOPHARM JAB. President Rodrigo Duterte receives his first shot of a Sinopharm Covid-19 vaccine on Monday night (May 3, 2021). Health Secretary Francisco Duque III administered the Sinopharm jab to the President. (Screenshot from Senator Bong Go’s Facebook livestream)
MANILA (Philippines News Agency) – President Rodrigo Duterte on Monday evening received his first shot of Covid-19 vaccine developed by Chinese state-owned pharmaceutical company Sinopharm.
Senator Christopher “Bong” Go went live on Facebook to show Duterte getting inoculated with Sinopharm. He was vaccinated by Health Secretary Francisco Duque III.
“I feel good. I have been expecting this shot vaccination a long time ago,” Duterte said in the livestream on Go’s Facebook page.
Duterte said it took his physician “a long time” to assess which vaccine brand to get.
“Sinopharm itong tinuturok sa akin (Sinopharm is what I am being inoculated with),” he added.
In a separate statement, Presidential Spokesperson Harry Roque said Duterte’s first dose was covered by the Compassionate Use Permit issued to the Presidential Security Group (PSG) hospital by the Food and Drug Administration (FDA).
In his previous speeches, Duterte has been vocal about his preference to receive a Sinopharm vaccine.
The Palace earlier denied rumors that Duterte has already been administered with a Covid-19 vaccine.
To recall, the PSG admitted to vaccinating some of its members with Sinopharm in September and October 2020.
Despite initially wanting it done in private, Duterte agreed to get vaccinated for Covid-19 in public to boost vaccine confidence among Filipinos.
He earlier said he was willing to waive his Covid-19 vaccine slot, but Malacañang said that his “personal conviction” may change when more vaccine supplies arrive in the country.
China approved the Sinopharm vaccine for general public use last December after the developer said the vaccine showed 79.34 percent efficacy based on an interim analysis of late-stage clinical trials.
On March 1, Roque said Sinopharm has filed an application for emergency use authorization (EUA) for its vaccines before the FDA of the Philippines.
The FDA said it has yet to receive all the required documents to carry out a full review of Sinopharm vaccines. (PNA)